If you look in the right-hand column of this blog, you'll see a new widget titled "Swift Observatory." That's because of a speedy satellite that is in need of some speedy attention.The Neil Gehrels Swift Observatory satellite was originally called the "Swift Gamma-Ray Burst Explorer" when NASA launched it in 2004. It has since been renamed to honor its longtime principle … [Read more...] about Swift Descending
Science
Trump’s dumb attempt to define sex
(When I started to write this post, I thought it would be a neat little think piece about science, policy, and meaning. But given how much more is going on, it now fells a bit academic. Nevertheless, I took the time to write it, so I might as well hit the Publish button...)Trump's anti-transgender executive order((The one from last week, not the new ones from this week. … [Read more...] about Trump’s dumb attempt to define sex
Covid Stats Dump – 2022-01-29
I usually tweet out Chicago-area Covid stats, but I thought I'd try something different today and post the information here on my blog. Most of the charts and maps are pulled from the wonderful Covid Act Now website. They take a lot of data and make it easy to understand.The national picture is looking extremely bleak, with the exception of Maryland, which is looking merely … [Read more...] about Covid Stats Dump – 2022-01-29
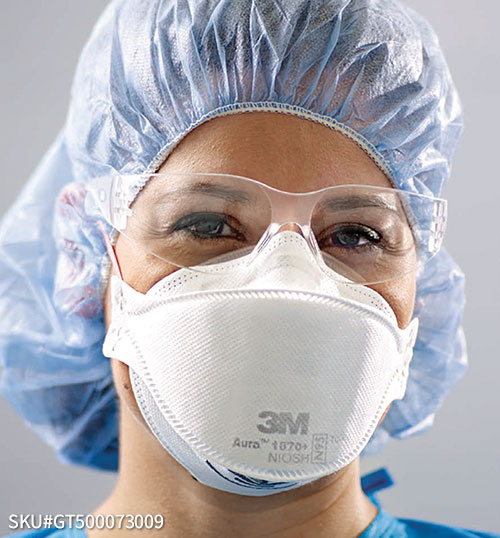
N95 or Bust!
This tweet reminded me that it's probably time for an update to my series of posts on COVID-19 masking:Public health messaging about this has been terrible. The truth is that not all masks are created equally, and some masks are much better than others. I think public health authorities have been reluctant to say this out loud for a number of reasons, such as not wanting … [Read more...] about N95 or Bust!
Living with Covid is like being lost in the forest
A few weeks ago, I ran across an opinion column by Ben Shapiro titled "When does the COVID-19 panic end?" I keep thinking about it, and how much it pissed me off, so I decided I might as well get it out of my system by writing about it.Two weeks to slow the spread.That was the original rationale for the lockdowns, masking and social distancing: Prevent transmission of … [Read more...] about Living with Covid is like being lost in the forest